13 July 2020
How do water softeners remove calcium and magnesium?
Calcium and magnesium are minerals (ions) found in hard water. Water softeners are used to remove those minerals from the hard water. Here’s how.
Water softeners contain two separate chambers: a brine tank and a resin tank. The resin tank is where the water softening takes place.
Water softeners make use of a process called ion exchange to remove hardness ions from the water. The water softener resin tank contains a bed of small beads that have sodium ions electrostatically attached to them. The water softener pumps hard water through this resin bed and the hardness ions are exchanged with the sodium ions. That is, the hardness ions will stick to the resin and displace the sodium ions which then end up in the softened water.
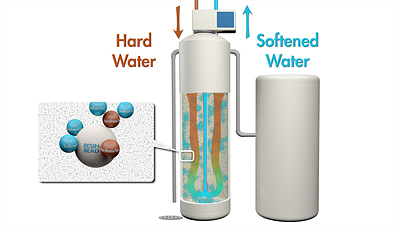 Over time the resin fills up with hardness ions and can no longer soften the water. At that point the water softener must regenerate. Salt brine from the brine tank is pumped into the resin and that salt brine washes away the hardness ions and replaces them with a fresh layer of sodium ions. The softener resumes softening more water.
Over time the resin fills up with hardness ions and can no longer soften the water. At that point the water softener must regenerate. Salt brine from the brine tank is pumped into the resin and that salt brine washes away the hardness ions and replaces them with a fresh layer of sodium ions. The softener resumes softening more water.
To learn more, check out our video.
Tagged with: ,
Cargill Salt considers our customers as partners and want to see our partners succeed. Cargill Salt has expanded our customer loyalty program, Partners in Profit (PIP) Advertising, to help WQA member customers grow their business, stay relevant and continue to thrive.
Water softener pellets are typically made from salt (sodium chloride) and are used in a water softener to remove hard water minerals, resulting in soft water.

